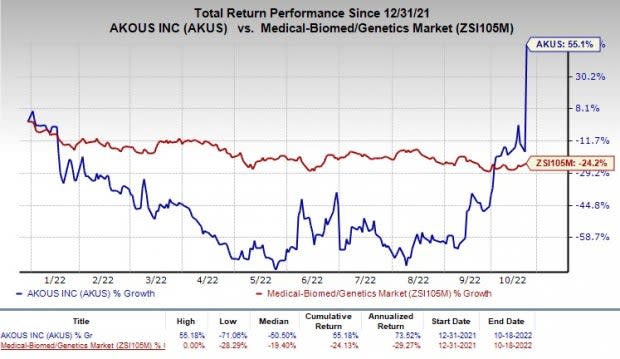
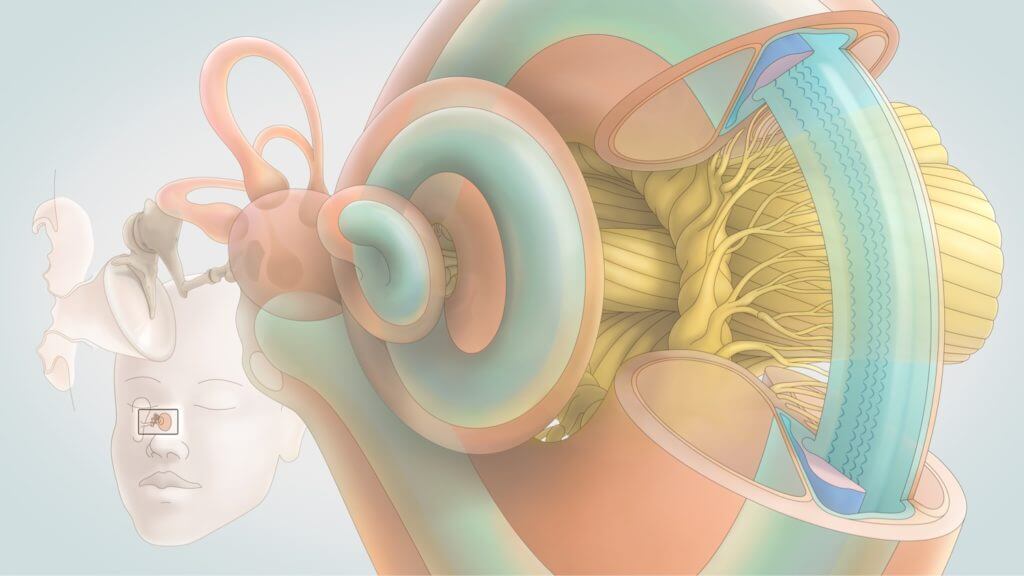
Akouos Receives FDA Clearance of its IND Application for Gene Therapy Hearing Loss Treatment, AK-OTOF | Hearing Health & Technology Matters

NEA on X: "🎉 Congratulations, Akouos! 👏🏽🤩🚀 Akouos to be acquired by @LillyPad: https://t.co/3qKl9lN3Wo https://t.co/UPMY69oh82" / X

Akouos Announces $7.5 Million Seed Round, Strategic License Agreement for Hearing Disorder Gene Therapy Platform | The Hearing Review